Vu Orthodontics - Orthodontics, Periodontics, and Implants
16027 Brookhurst St., Ste. K
Fountain Valley, CA 92708
ph: 714-775-0100
fax: 714-463-2205
drhungvu
- Home
- COVID-19 Notice
- CBCT
- CBCT & Chiropractic
- Biomechanics
- Services
- About Us
- Meet the Doctors
- Contact Us
- Cases
- Testimonials
- Emergency
- Office
- Ortho TADs
- Class II ("overbite")
- Class III ("underbite")
- Extremely Crowded
- Oral Surgery & Periodontics for GP
- Implants
- Medications
- Phlebotomy, PRF, PRP & SB
- Phase I (Pre-Teen Orthodontics)
- Adult Orthodontics
- Lasers
- Impacted Canines
- Accelerated Orthodontics
- Impacted Wisdom Teeth
- Referral Form
- Study Club
- Journal Articles
Phlebotomy, PRP, PRF
Introduction
This writing presents the current state of knowledge regarding PRF (Platelet Rich Fibrin), PRP (Platelet Rich Plasma) and SB (Sticky Bone) in oral/periodontal surgeries and implantology. SB is a blob of bone particulates that stick together and include lots of growth factors.
Although SB contains lots of growth factors which are good for bone growth, it may cause inflammation. If so, the consequence is delayed healing.
The aim is to provide readers with the necessary understanding in producing PRF and SB in good quantity and quality so they can be utilized for better clinical practice. This work will be an asset for general dentists and dental specialists who are involved in treating patients who need oral/periodontal surgeries and implants. It will also be a useful source of information for residents of certain dental specialties who are seeking information on PRF and SB.
PRF and SB are important in socket graft (for ridge preservation), Guided Bone Regeneration (GBR) which includes vertical and horizontal ridge augmentation, and Guided Tissue Regeneration (GTR).
The mechanism that produces SB is explained as follows. First, fibrinogen (I) must be obtained. But the trick is how to get the plasma that contains fibrinogen before it turns into fibrin (Ia). Either a white-cap plastic tube or a yellow-cap plastic tube, without any coating, is used to obtain this plasma. When this plasma is mixed with bone-graft particulates, they will coagulate iff (if and only if) there is thrombin (IIa). And thrombin converts fibrinogen into fibrin. When the PRF is squeezed/pressed, it yields a liquid which contains thrombin. This liquid is called by some as Autologous Fibrin Glue (AFG). In conclusion, SB is made by mixing bone particulates with fibrinogen and thrombin.
Note that Sohn et al (2015) used the terms Autologous Fibrin Glue (AFG) and exudate to refer to this liquid. But according to Merriam-Webster, exudate is defined as "the material composed of serum, fibrin, and white blood cells that escapes from blood vessels..."
In addition, according to Wikipedia, "Thrombin ... is a serine protease, an enzyme ... Prothrombin (coagulation factor II) is proteolytically cleaved to form thrombin in the clotting process. Thrombin in turn acts as a serine protease that converts soluble fibrinogen into insoluble strands of fibrin, as well as catalyzing many other coagulation-related reactions."
What type of tube should be used to obtain PRF, red-cap glass tube or red-cap plastic tube? The answer is that it would better to use the red-cap glass tube instead of the red-cap plastic tube (with silica coating). The reason is common glass is silica-based glass, and silica (aka silicon dioxide or quartz) is a coagulating agent. Thus, glass tube already has silica built-in. If the plastic tube is used, the silica coating would be mixed with the bone graft material. Clearly, having silica in the bone graft is harmful.
In the pursuit of perfection in phlebotomy (venipuncture), for the purpose of utilizing PRF and PRP in periodontal surgeries and implantology, the author has become a licensed phlebotomist. The author routinely uses PRF for surgical procedures in periondontics and implantology, to enhance healing.
The most common location for venipuncture is the antecubital area of the arm, where the following veins are located: median cubital (also called medial cubital), basilic, and cephalic. They are most prominent and usually superficial to the skin (Fig. 1)
Fig. 1. Median cubital vein (middle), cephalic vein (left), basilic vein (right)
Median cubital vein is often the best choice for venipuncture when it is visible and relatively large. This vein lies over the cubital fossa, and it is an anastomosis between the basilic and cephalic veins.
Median cubital vein is the best because it does not roll (stable) whereas basilic and cephalic veins may roll (unstable).
If the median cubital vein cannot be located (because it is so deep that cannot be seen or palpated), the alternative is the cephalic vein, and the next alternative is the basilic vein. As an example, Fig.2 shows the venipuncture site in the cephalic vein, and the butterfly needle was used.
Fig. 2. Cephalic vein and a butterfly needle. The alcohol pad's corner was used to aid the entering (location and orientation) of the needle.
After locating the vein by palpation or visual inspection, the site is wiped with an alcohol pad, in an outward circular motion. It is important to point out that this kind of motion is used for better infection control. Unfortunately, some health professionals may not follow this practice. Next, the corner of the alcohol pad was used to mark the point where the needle would enter -- a bit from it.
It is typical that some health professionals may re-palpate the vein after disinfecting the site since the exact location cannot be identified without any marking. Doing so will contaminate the site unless surgical gloves were used, but nobody uses surgical gloves in phlebotomy.
It should be noted that a typical phlebotomist would not know how to produce PRF and PRP. The function of a phlebotomist is mainly drawing the blood -- only for testing purposes.
The two barriers for a typical general dentist or dental specialist are: 1. not being able to perform a successful venipuncture for patients with poor vein conditions, and 2. not being able to produce PRF and SB of high quantity and quality for different centrifuge machines (with different parameters) and different blood collection tubes.
A typical dentist would learn about the PRF & SB either from a one or two-day course or some training program and then purchase a centrifuge machine (Fig. 2), expecting to be able to use the technique of PRF & SB.
Fig. 2 The rotating part of a centrifuge machine
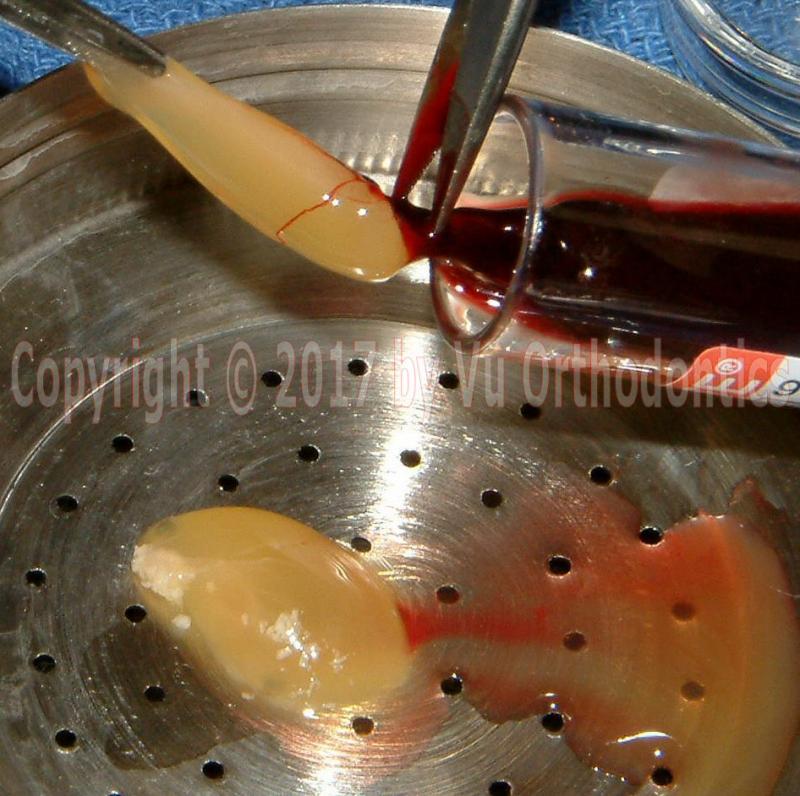
Due to their roles in wound healing and regeneration, PRF and SB, shown in Figs. 3-5, have been utilized in periodontics, and implantology, even in modern endodontics. They have also been used in medicine, including dermatology. Typically, PRF may be used as a membrane, but it is weak. The timing is critical for making SB. Although PRF and SB have been used for a long time, there is still much confusion in many aspects.
Fig. 3 PRF (in red-cap tube) and plasma that contains fibrinogen (in white-cap tube)
Fig. 4 PRF (Platelet-Rich Fibrin)
Fig. 5 SB (Sticky Bone)
The main barriers for general dentists and dental specialists (hereinafter "the dentists") in utilizing the technique of using PRF and SB are drawing the blood and obtain PRF an SB in both good quality and quantity.
An easily detected vein is the one that can be seen readily or can be palpated. If it can be seen easily, but it is too small, then it is bad. In addition, a poor vein can be flat and buried deep in the soft tissue so it cannot be palpated. A good/easy vein for phlebotomy is plump, relatively large, and does not roll/move.
A successful venipuncture is defined as placing the needle correctly inside the vein and being able to collect a good amount of blood. In some instances, a small amount of blood is collected, but after that, the blood is no longer flowing.
For some patients with a poor vein condition, even when the needle is placed correctly inside the vein, there is no blood flow. The vein can be collapsed when the vacuum from the collection tube starts to exert a negative pressure on the vein.
For skilled phlebotomists, drawing the blood from most patients is relatively easy. It is natural because that's what they profess, and they perform the procedure many times during any working day. But, even for them, sometimes, it can be a humbling experience when a patient presents with an extremely poor vein condition.
Typically, other health professionals (dentists, medical doctors, nurses) do not possess the skill set of an experienced phlebotomist in blood drawing.
But unlike a typical injection (e.g., inferior alveolar nerve block) that any dentists perform on a patient, blood drawing can be embarrassing if it fails. It would be not obvious if the injection fails or is less than ideal. But a failed blood drawing would be obvious to both the patient and the dentist.
A fundamental rule in phlebotomy is that after two unsuccessful attempts, a phlebotomist should ask another co-worker to take over. It is not fair to the patient to be poked over and over.
If a phlebotomist (in a phlebotomy lab) or a nurse (in a health facility) has difficulty in drawing the blood, there would always be a more experienced phlebotomist or nurse to help out.
But in a typical dental office, if the dentist is the only one who can draw the blood, and if he/she fails and loses confidence, the centrifuge machine might be just sitting somewhere and collecting dust.
Fundamentals
About 7-8% of our total body weight is whole blood (WB) which contains RBCs (red blood cells), WBCs (white blood cells), and plasma. RBCs are about 45% and plasma is about 55%. The combined WBCs and platelets is about 1%.
To check for anemia, the hematocrit test is for the volume of RBCs in the WB. Normal hematocrit, on the average, is 45%. It is about 47% for men and 42% for women.
To check for bleeding/coagulation problems, before a major surgery, the following 3 tests are needed:
1. PT (Prothrombin Time) / INR (International Normalized Ratio). Depending on the analytical method used, the reference range for PT is 12–13 s. The INR is the ratio of a patient's PT to a control sample, raised to the power of the ISI (International Sensitivity Index) value, for the analytical system used. In absence of anticoagulation medication (e.g., warfarin), the normal range for INR is 0.8–1.2. But with anticoagulant therapy, the target range for INR is 2-3. (Ref: Prothrombin time, Wikipedia)
2. PTT (Partial Thromboplastin Time)
3. Platelets count -- from CBC (Compete Blood Count) test
Approximately, after donating a unit of blood, about 350-450 mL, the body replaces plasma within 2-3 days, but it takes about 4-8 weeks to replace the RBCs.
An average person has about 5 L of blood, so each blood-donation unit is a little less than 10%.
A matured RBC (erythrocyte) lives (in blood circulation) for about 120 days. Bone marrow makes RBCs whose genesis cannot be equated to their lifespan.
Platelet (thrombocyte), the smallest of the blood cells, is about 1/20 the size of the RBC.
The immature RBC is called reticulocyte, and the mature RBC is called erythrocyte.
The WBCs, also called leukocytes or leucocytes, are of 5 types: neutrophils (40%-60%), lymphocytes (20%-40%), monocytes (2%-8%), eosinophils (1%-4%), and basophils (0.5%-1%). A mnemonic device is given as Never Let Monkey Eat Banana (N -> L -> M -> E -> B).
Some Collection Tubes in Phlebotomy
A phlebotomy lab would use many different tubes for a variety of tests. The tubes are signified by the color of the cap, e.g., a red-capped tube which is simply called a red tube, for short.
The two most common test tubes are SST (Serum Test Tube, also called tiger top) and lavender. The next common test tube is light blue. There are many other test tubes: red, royal blue with red stripe, light green, dark green, royal blue with blue stripe, pink/pearl, tan, gray, and others.
The SST tube is also used to detect a critical biomarker for cancer: CEA (CarcinoEmbryonic Antigen) which is the protein that appears in the blood of some patients who have cancers, such as liver, large intestine (colon and rectal cancer), pancreas, breast, ovary, and lung.
A pure serum is required in many fundamental tests in medicine. And an SST collection tube is typically used to produce serum.
Collection Tubes, PRF & SB
For the purposes of making PRF & SB, two types of collection tubes should be used:
1. Yellow-top plastic tube (for making SB). This tube must have no additive or coating. It is a mistake of using the yellow-tube plastic tube with acid citrate dextrose (ACD) as an additive for the purpose of making SB since ACD (also called Solution A) is an anticoagulant. The reason that we want no anticoagulant is because it would prevent coagulation which is what we need to make SB. As an alternative, a white-top plastic tube without any additive (or any coating) can also be used.
2. Red-top glass tube (for making PRF and SB). The glass tube needs no silica coating (as clot activator) on the inner wall because the glass itself is silica (for coagulation, to make PRF). As an alternative, one can also use a red-top plastic tube, but this tube must have silica coating (for coagulation). This coating can come off and mix with PRF, which is harmful. Note that the red-top glass tube is slightly more expensive than its plastic cousin.
After being spun, the yellow-top tube will yield autologous fibrin glue (AFG) which is in a liquid form, which must be collected right away, to prevent it from being coagulated. This AFG contains fibrinogen which is a precursor of fibrin.
Similarly, after being spun, the red-top tube will yield the PRF which is in a semi-solid gel form. When the PRF is being squeezed, it yields a liquid which contains thrombin.
To make SB, we first mix the AFG to the bone-graft particulates (or particles), then we add the thrombin to the mix. Fibrin will activate the fibrinogen in the AFG to become fibrin. Thus, SB is made.
More on Collection Tubes
The white tube also has three layers: the bottom is RBCs, the middle one is the thin buffy coat (BC), and the top is plasma. The buffy coat (so called because of the buff color which is light yellow) contains the leukocytes or WBCs (white blood cells) and platelets (thrombocytes). Within this top portion, next to the BC is the PRP (platelet-rich plasma) followed by the PPP (platelet-poor plasma).
It should be noted that this is a result of a single spin.
Other authors claim that PRP must be obtained from a double-spin procedure. After the first (soft) spin, the bottom part of the collection tube contains RBCs since they are the heaviest, the next thin layer is the BC which contains WBCs and platelets, and the top part is plasma. Then, the RBCs are discarded. After the second (hard) spin, the WBCs stay at the bottom as a thin layer, the next portion is PRP, and the top part is PPP.
After the blood collection, the tubes must be spun immediately. If the condition is ideal, we should get a high-quality amount of PRF from a red tube (right after spinning). We could also get some PRF from a white tube if we let it sit for a while (after spinning, approximately 15-30 minutes), but the quality and quantity of this PRF would be poor.
Platelets and Clotting Factors
In periodontal surgery and regeneration therapy, growth factors are critical. Thus, platelets are what we need and want. But clotting factors are also important in surgery because we need coagulation.
Platelets secrete the following growth factors: PDGF (platelet-derived growth factor), VEGF (vascular endothelial growth factor), and TGF (transforming growth factor). And these growth factors promote wound healing by cellular proliferation.
Clotting factors (also called coagulation factors) are Factors I (Fibrinogen), Ia (Fibrin), II (Prothrombin), IIa (Thrombin), III, IIIa, IV, IVa, V, Va, VI, VIa, VII, VIIa, VIII, VIIIa, IX, IXa, X, Xa, XI, XIa, XII, XIIa, XIII, and XIIIa. These factors respond in a complex cascade to finally form a stable fibrin clot (or strands).
After the red tubes are spun by a working centrifuge machine with proper settings, they would have three different layers: the bottom is the RBCs (red blood cells), the middle is PRF (platelet-rich fibrin, a gel), and the top. What do we call this top layer?
Some people call this top layer as PPP (platelet-poor plasma). By definition, that would be correct if it has some platelets since most platelets are assumed to be in the PRF.
Also, by definition, plasma is serum plus clotting factors. In other words, serum is the liquid portion of plasma after the clotting factors are excluded.
Unfortunately, there is some confusion in the literature regarding plasma and serum.
If most clotting factors are in the PRF, and only some clotting factors are in the top layer, then this layer should be called CFPP (Clotting-Factor Poor Plasma) -- not PPP (platelet poor plasma).
This liquid would be called PPP (platelet poor plasma, a type of plasma) if it contained all the clotting factors and if PRF contained no clotting factor. But this is not the case in reality.
In summary, platelets contain growth factors, whereas plasma contains clotting factors. And both platelets and clotting factors are important in dental surgeries.
Highlights of Coagulation Cascade
The clotting or coagulation cascade occurs through two separate pathways that interact:
1. intrinsic (contact activation) pathway
2. extrinsic (tissue factor) pathway
The intrinsic pathway involves Factors XII, XI, IX, VIII. It is activated by internal trauma (inside the blood vessels). For example, it is activated by platelets. An injury to a tissue surface will activate Factor XII to become Factor XIIa. The intrinsic pathway is slower than the extrinsic pathway.
The extrinsic pathway involves Factor VII. It is activated by external trauma that cuts blood vessels. The trauma activates Factor VII to become VIIa. The extrinsic pathway is faster than the intrinsic pathway.
After the complex coagulation cascade, Factors Va and Xa activate prothrombin (Factor II) to become thrombin (Factor IIa).
In turn, thrombin (Factor IIa) activates fibrinogen (Factor I) to becomes fibrin (Factor Ia) which finally becomes a cross-linked fibrin clot when activated by Factor XIIIa.
Clinical Significance
PRF can be obtained at a low cost, provided the dentist has the skill of blood drawing and a good working knowledge of centrifuge machine and collection tubes. A question often raised is should we need to place a collagen membrane on top of the PRF membrane? It turns out to be, it is better to use the PRF membrane alone. From limited observations of the author, PRF and collagen membranes may interact and cause some form of degradation, and it could be hydrolysis.
In addition, PRF membrane has adhesion property so it adheres to the incision line (thus the graft is better protected) where collagen membranes would not.
If the surgical site is well sutured, the use of PRF membrane -- without collagen membrane -- will give rapid soft tissue healing.
At this point, there is no consensus that PRF/PRP would enhance bone healing/growth.
Mechanics
Which term is correct: centrifugal force or centripetal force?
Centrifugal means leaving the center, whereas centripetal means seeking the center.
An object with a momentum will continue to move in a straight line, in the absence of any applied force.
Momentum is the product of mass times velocity vector. A vector has 2 properties: magnitude and direction. Speed is the magnitude of a velocity vector.
To maintain an object moving in a circle, there must be an applied force, and this force is called centripetal force, the object is seeking the center. Without an applied force, the object will continue to move in a straight line, thus, leaving the center. Centrifugal force does not exist, but many people are confused about these two terms: centrifugal force and centripetal force.
It is called "centrifuge machine," but one should know that when an object is spun, it tries to leave the center, hence the term centrifugal. Centrifugal force actually does not exist, in this application.
RCF
Does the acronym RCF stand for Relative Centrifugal Force or Relative Centripetal Force? According to the explanation above, it should be Relative Centripetal Force.
Again, centrifugal force does not exist.
Optimal settings
In a strict sense, optimal means the best which may be elusive. In clinical practice, sub-optimal may be what we can only hope for.
According to some authors, S-PRF (Standard Platelet Rich Fibrin) is obtained at 2700 rpm (revolution per minute) for 12 minutes, whereas A-PRF (Advanced Platelet Rich Fibrin) is at 1500 rpm for 14 minutes. Moreover, the A-PRF + (Advanced Platelet-Rich Fibrin Plus) is obtained at even slower spinning speed.
But rpm is not a unique setting for different centrifuge machines because they have different radii. Thus, RCF should be used instead.
According to BD (the manufacturer of the blood collection tubes), the recommended settings for the gel tubes (in fixed-angle centrifuge) are 1000-1300 RCF (g) for 15 minutes. These settings equate to 2900-3300 rpm on a Champion F-33D.
Gel tube is a tube which contains a gel as a separator to produce serum -- not PRF.
Calculating RCF from known omega (in rpm) and R (radius, in cm):
General formula:
RCF = 11.2*R*(omega/1000)^2
where:
RCF = Rotational Centripetal Force (in g)
g (gravity) = 32.17 ft/s^2 or 9.81 m/s^2
omega = rotational speed, in rpm (revolution per minute)
R = radius, measured from the center of rotation to the bottom of the collection tube, in cm
Example: For our current centrifuge machine, Champion F-33D, the values are
omega = 2700 rpm; R = 11.07 cm
Thus,
RCF = 11.2*11.07*(2700/1000)^2 = 904 g
Some authors suggest that the better RCF for producing good PRF (in quantity and quality) should be around 400 g or 450 g. Thus, 904 g or 2700 rpm is excessive.
The RPM can be obtained from a selected RCF by the following expression,
omega = 1000*sqrt(RCF/(11.2*11.07))
Thus,
omega = 1796 rpm if RCF = 400 g
omega = 1905 rpm if RCF = 450 g
We have good results for PRF (good quality and quantity) with the following settings: omega =1800 rpm, time = 8 minutes.
At high speed, the tubes will be rotated with significant vibrations if there is an imbalance. And the results will be poor.
For this machine (Champion F-33D), the configuration must be either 4 tubes or 8 tubes, for good balancing. The configuration of either 2 tubes or 6 tubes is bad, for balancing.
Note that for a particular setting, the results are different when the tubes (e.g. 4) are full and when they are significantly less.
Literature Review & References
1. According to Ghanaati et al, "Advanced Platelet-Rich Fibrin: A New Concept for Cell-Based Tissue Engineering by Means of Inflammatory Cells," Journal of Oral Implantology, Vol. XL, No. 6, 2014 (DOI: 10.1563/aaid-joi-D-14-00138):
"...protocols for standard platelet-rich fibrin (S-PRF) (2700 rpm, 12 minutes) and advanced platelet-rich fibrin (APRF)(1500 rpm, 14 minutes) were compared to establish by histological cell detection and histomorphometric measurement of cell distribution the effects of the centrifugal force (speed and time) on the distribution of cells relevant for wound healing and tissue regeneration. Immunohistochemistry for monocytes, T, and B-lymphocytes, neutrophilic granulocytes, CD34-positive stem cells, and platelets was performed on clots produced from four different human donors. Platelets were detected throughout the clot in both groups, although in the A-PRF group, more platelets were found in the distal part, away from the buffy coat (BC). T- and B-lymphocytes, stem cells, and monocytes were detected in the surroundings of the BC in both groups.Decreasing the rpm while increasing the centrifugation time in the A-PRF group gave an enhanced presence of neutrophilic granulocytes in the distal part of the clot. In the S-PRF group, neutrophils were found mostly at the red blood cell (RBC)-BC interface. Neutrophilic granulocytes contribute to monocyte differentiation into macrophages. Accordingly, a higher presence of these cells might be able to influence the differentiation of host macrophages and macrophages within the clot after implantation. Thus, A-PRF might influence bone and soft tissue regeneration, especially through the presence of monocytes/macrophages and their growth factors. The relevance and feasibility of this tissue-engineering concept have to be proven through in vivo studies."
2. According to Ehrenfest et al, "The impact of the centrifuge characteristics and centrifugation protocols on the cells, growth factors and fibrin architecture of a Leukocyte- and Platelet-Rich Fibrin (L-PRF) clot and membrane. Part 1." POSEIDO. 2014;2(2):
"The A-PRF centrifuge (Advanced Platelet-Rich Fibrin) is an interesting case, as it was suggested to be used with a very low speed (1300 rpm) in order to produce a PRF-like clot called A-PRF. This is actually a quite rare approach, as a too low speed does not allow a good separation of the blood components and the activation of leukocytes.
...
In theory, the best configuration for L-PRF would be to have a reasonable speed (around 2700 rpm) for adequate blood separation, and no vibration or resonance to protect the cell content [9], as the adequate collection of the leukocyte appeared as an important parameter for the clinical effect of these technologies [32,33]. Therefore, A-PRF could serve as an interesting example to compare with the original L-PRF, and to illustrate the impact of speed and vibrations on the final aspect and content of a PRF clot."
3. Dong-Seok Sohn, Bingzhen Huang, Jin Kim, Eric Park, Charles C. Park, Utilization of Autologous Concentrated Growth Factors (CGF) Enriched Bone Graft Matrix (Sticky Bone) and CGF-Enriched Fibrin Membrane in Implant Dentistry, The Journal of Implant & Advanced Clinical Dentistry Volume 7, No. 10, December 2015, pp 11-29
4. Prothrombin time, Wikipedia
© Copyright 2007-2014, Vu Orthodontics. All rights reserved.
Serving communities: Orange County, Fountain Valley, Huntington Beach, Westminster, Garden Grove, Anaheim, Santa Ana, Buena Park, Tustin, Costa Mesa, Newport Beach, Irvine, Stanton, Cypress, Seal Beach, Los Alamitos, Fullerton, La Habra, Brea, Yorba Linda, Placentia, Villa Park, Lake Forest, Mission Viejo, Laguna Beach, Laguna Niguel, Laguna Hills; Los Angeles, Long Beach...
16027 Brookhurst St., Ste. K
Fountain Valley, CA 92708
ph: 714-775-0100
fax: 714-463-2205
drhungvu